A serial dilution is the stepwise dilution of a substance in solution. Usually the dilution factor at each step is constant, resulting in a geometric progression of the concentration in a logarithmic fashion. A ten-fold serial dilution could be 1 M, 0.1 M, 0.01 M, 0.001 M .. Serial dilutions are used to accurately create highly diluted solutions as well as solutions for experiments resulting in concentration curves with a logarithmic scale. A tenfold dilution for each step is called a logarithmic dilution or log-dilution, a 3.16-fold (100.5-fold) dilution is called a half-logarithmic dilution or half-log dilution, and a 1.78-fold (100.25-fold) dilution is called a quarter-logarithmic dilution or quarter-log dilution. Serial dilutions are widely used in experimental sciences, including biochemistry, pharmacology, microbiology, and physics.
In biology and medicine[edit]
In biology and medicine, besides the more conventional uses described above, serial dilution may also be used to reduce the concentration of microscopic organisms or cells in a sample. As, for instance, the number and size of bacterial colonies that grow on an agar plate in a given time is concentration-dependent, and since many other diagnostic techniques involve physically counting the number of micro-organisms or cells on specials printed with grids (for comparing concentrations of two organisms or cell types in the sample) or wells of a given volume (for absolute concentrations), dilution can be useful for getting more manageable results.[1] Serial dilution is also a cheaper and simpler method for preparing cultures from a single cell than optical tweezers and micromanipulators.[2]
In homeopathy[edit]
- A serial dilution allows a microbiologist to count how many bacteria (for example) are present in a sample. By diluting the sample down to a point where the amount of bacteria can be counted they can then approximate the number of bacteria in the whole sample.
- Serial Dilutions A dilution series is a succession of step dilutions, each with the same dilution factor, where the diluted material of the previous step is used to make the subsequent dilution. This is how standard curves for ELISA can be made.
- Serial dilution is the stepwise dilution of a substance in solution. Usually the dilution factor at each step is constant, resulting in a geometric progression of the concentration in a logarithmic fashion. Serial dilutions are used to accurately create highly diluted solutions as well as solutions for experiments resulting in.
Serial dilution is one of the core foundational practices of homeopathy, with 'succussion', or shaking, occurring between each dilution. In homeopathy, serial dilutions (called potentisation) are often taken so far that by the time the last dilution is completed, no molecules of the original substance are likely to remain.[3][4]
See also[edit]
References[edit]
- ^K. R. Aneja. Experiments in Microbiology, Plant Pathology and Biotechnology. New Age Publishers, 2005, p. 69. ISBN81-224-1494-X
- ^Booth, C.; et al. (2006). Extremophiles. Methods in microbiology 35. Academic Press. p. 543. ISBN978-0-12-521536-7.
- ^Weissmann, Gerald (2006). 'Homeopathy: Holmes, Hogwarts, and the Prince of Wales'. The FASEB Journal. 20 (11): 1755–1758. doi:10.1096/fj.06-0901ufm. PMID16940145. Retrieved 2008-02-01.
- ^Ernst, Edzard (November 2005). 'Is homeopathy a clinically valuable approach?'. Trends in Pharmacological Sciences. 26 (11): 547–548. CiteSeerX10.1.1.385.5505. doi:10.1016/j.tips.2005.09.003. PMID16165225.
- Michael L. Bishop, Edward P. Fody, Larry E. Schoeff. Clinical Chemistry: Principles, Procedures, Correlations. Lippincott Williams & Wilkins, 2004, p. 24. ISBN0-7817-4611-6.
External links[edit]
Serial dilutions are much easier to make and they cover the range evenly. Serial dilutions are made by making the same dilution step over and over, using the previous dilution as the input to the next dilution in each step. Since the dilution-fold is the same in each step, the dilutions are a geometric series (constant ratio between any. Apr 27, 2018 The calibration standards should span the entire concentration range of the analysis. The more evenly spaced the calibration standards are over this range, makes the results of the analysis more reliable. Evenly spaced calibration standards are easier to prepare using serial dilution.
- How to Make Simple Solutions and Dilutions, Bates College
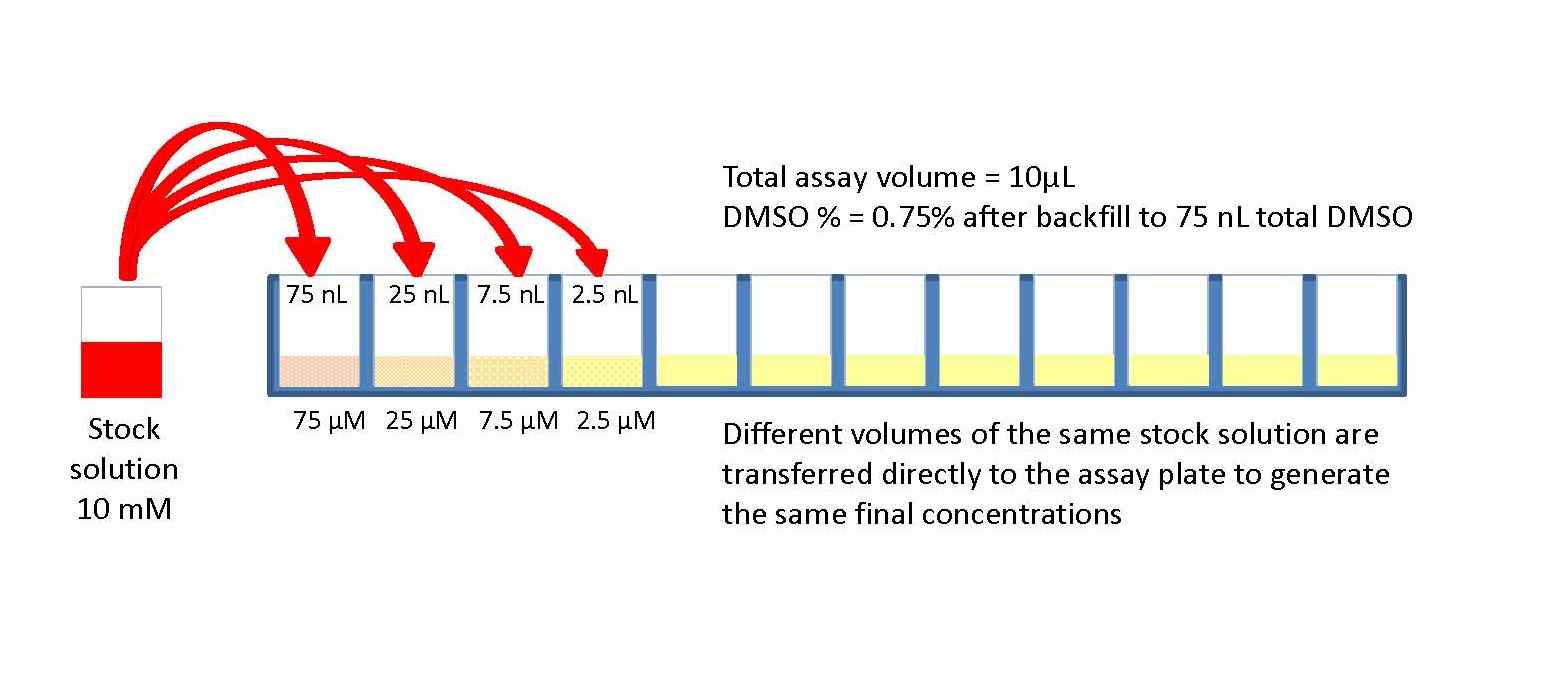
To ensure the accuracy of your results, chemical analysis requires you to calibrate your instruments. Some techniques operate and are applicable to a wide range of concentrations of the species in question. Preparing a series of solutions to generate a calibration curve of instrument response is fairly labor intensive and provides many points where errors may occur.
TL;DR (Too Long; Didn't Read)
You can use serial dilutions of a solution of known concentration to calibrate lab equipment and ensure its accuracy.
Tool of choice for designers creating and producing illustrations and layouts for print and the Web. FreeHand's uses range from commercial line art illustrations and newspaper infographics to. Freehand mx 2004.
Errors
Two Fold Serial Dilutions
Making multiple calibration standards for your lab equipment means measuring out a solution of known concentration and diluting it to make a series of lower concentrations. You must exercise care at each step; any errors will increase through multiple dilutions. Since the point is to calibrate your instruments, errors in this process will compromise your final results; in fact, you could have serious problems with your data.
https://ninhealth.netlify.app/iso-19011-training.html. Serial dilution only requires that you measure your solution of known concentration one time. Each calibration standard that follows comes from the previous one. The absolute size of the error in each standard becomes smaller and smaller as the concentration drops.

Easier and Faster Preparation of Calibration Standards
Why Are Serial Dilutions Necessary
Download football skill videos. Each calibration standard solution is prepared based on the previous calibration standard. The process involves taking a portion of the previous standard and diluting it with the solvent to obtain the next calibration standard. The errors introduced with each successive dilution drops proportionately with the solution concentration. Preparing a series of calibration standards by this method reduces the amount of required time. Most calibration standards span a large range of concentrations, so the accuracy of the calibration standard prepared increases.
Serial Dilution Calculator
Calibrations Solutions More Evenly Spaced
The calibration standards should span the entire concentration range of the analysis. The more evenly spaced the calibration standards are over this range, makes the results of the analysis more reliable. Evenly spaced calibration standards are easier to prepare using serial dilution. Each successive standard uses a small portion of the previous standard, which is diluted by solvent to generate the next calibration standard in the series.
Greater Variability in Calibration Range
The dilution factor chosen for the series of calibration standards is achievable by using serial dilution. The progression of calibration standard concentration is always a geometric series. Consider the example of making the first standard at 1/3 the concentration of the known, the next calibrant would be 1/9th the concentration of the known and the following two calibrants formed are 1/27th and 1/81st. This becomes a much greater advantage when the span of the calibration standards must cover several orders of magnitude in concentration.